Gas Pressure Is a Result of Which of the Following
1 Answer Nam D. What would increase the pressure of a gas in a container.
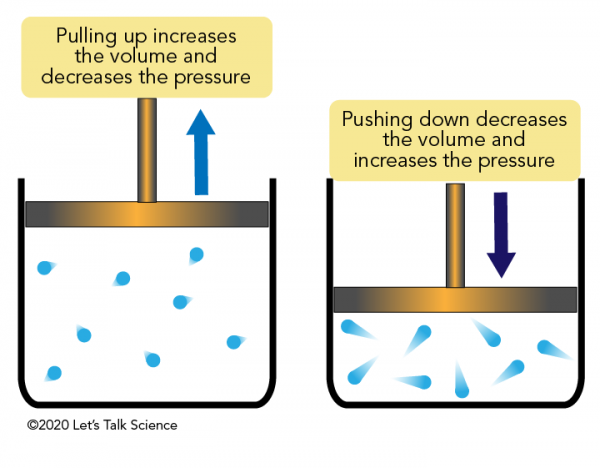
Boyle S Law Let S Talk Science
Apr 6 2018 0312 moles of water.

. Although the force of each collision is very small any surface of appreciable area experiences a large number of collisions in a short time which can result in a high pressure. Part A Option B is correct Gas pressure is the result of the collision of the gas particles with each other i. A smaller container exerts more pressure on the gas.
Molecules are slower at a cold temperature resulting in a lower gas pressure. According to Charless Law at constant pressure as the temperature T of a gas increases the volume V increases and vice versa. View the full answer.
Hence pressure of gas is the. Because of this increasing the number of air particles increases the number of. At constant temperature the volume of the container that a sample of nitrogen gas is in is doubled.
Use the gas laws and kinetic theory to relate the pressure volume and temperature of a gas to the motion of the molecules within it for National 5 Physics. X collisions of gas particles with each other a. Volume and pressure in gases the gas laws Boyles law.
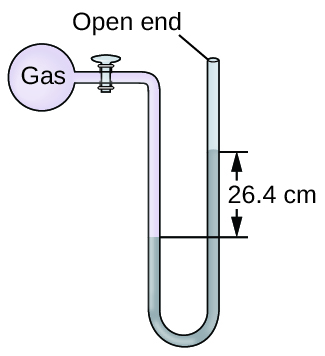
At constant volume and temperature the pressure of a gas is directly related. The device shown in the image is a closed-end _____ used for measuring gas pressure in an experiment. As a result the pressure of the nitrogen gas is halved.
The collisions between the particles are elastic. 4 Gas pressure is a result ofwhich of the following. The gas is at high pressure.
The correct option is B Molecular speed and mass. Increasing the amount of gas. The amount of nitrogen gas is unchanged in this process.
Although the force of each collision is very small any surface of appreciable area experiences a large number of collisions in a short time which can result in a high pressure. At constant temperature and pressure the volume of gas is directly related to its number of moles c. Compressibility of gas particles Phase diagram t.
At constant volume and number of moles the temperature of a gas is directly related to its pressure d. 24 Gas pressure is a result of which of the following. Comprehensive results include Reynolds number friction factor friction losses fitting losses fluid velocities and more.
Which of the following is an assumption of the kinetic model of an ideal gas. Gas pressure is caused by the force exerted by gas molecules colliding with the surfaces of objects. We got the balanced equation.
Decreasing the volume of a gas increases the pressure of the gas. In the closed-ended design shown if a gas is present in the flask the gas will exert a pressure on the mercury column closest to it causing the column on the left to be _____than the column on the right. An example of this is when a.
Gas pressure is caused by the force exerted by gas molecules colliding with the surfaces of objects link. Oxygen or compound molecules made from a variety of atoms eg. This is an example of.
The resistance of gas particles to flow d. Inside the balloon the gas particles collide with the. The correct answer is D The volume of the gas will increase.
At constant pressure and volume the temperature of a gas is directly related to its number of moles b. The pressure of the gas is the force that the gas exerts on the container boundaries. V1 T1 V2 T2.
Collisions of particles with the inside walls of the tire cause the pressure that is exerted by the enclosed gas. Any of the following. A pure gas may be made up of individual atoms eg.
Gas pressure is caused by the force exerted by gas molecules colliding with the surfaces of objects Figure 1. This is observed according to the formula. Answer in units of mol.
A noble gas like neon elemental molecules made from one type of atom eg. Gas is one of the four fundamental states of matter the others being solid liquid and plasma. -In an open-end manometer the gas pressure pushes on the Hg surface on one arm of the U tube and atmospheric pressure gas pushes on the other.
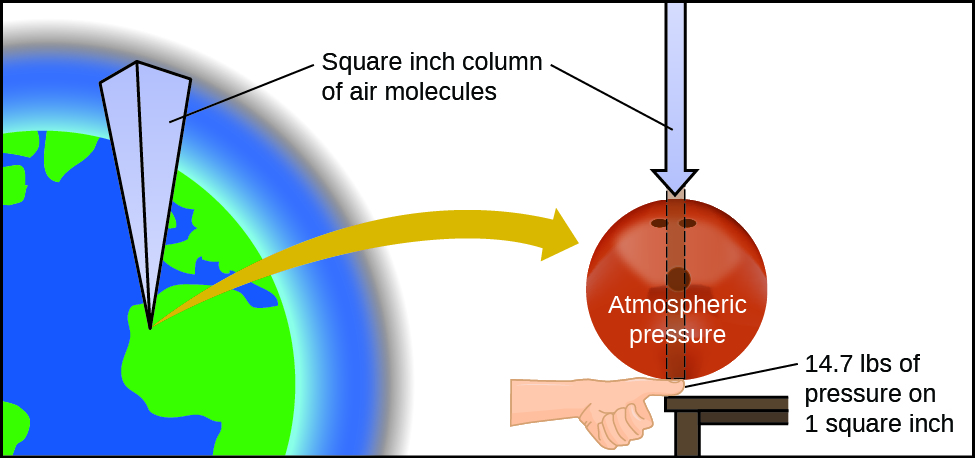
Chemical Engineering questions and answers. A collisions of gas particles with each other B expansion of gas particles in an open container C the resistance of gas particles to flow D collisions of gas particles with the walls of the container E compressibility of gas particles. Earths atmosphere exerts pressure because gravity acts on the huge number of gas particles contained in the atmosphere holding it in place.
When a container is heated the molecules move faster due to the extra energy. Gas pressure is the pressure that results from collisions of gas particles with an object. -The gas pressure is directly related to the height of a column of mercury it produces in a U tube.
Carbon dioxideA gas mixture such as air contains a variety of pure gases. Although the force of each collision is very small any surface of appreciable area experiences a large number of collisions in a short time which can result in a high pressure. Force is nothing but the product of mass and acceleration of molecule.
O expansion of gos porticles in an open container collisions of gos particles with each other the resistance of gas particles to flow collisions of gas particles with the walls of the container compressibility of gas porticles O Gay. This results in a higher gas pressure. AAvogadros Law BBoyles Law CGay-Lussacs Law DDaltons Law ECharless Law.
The energy of the. Acetylene gas C2H2 is produced as a result of the reaction CaC2s 2 H2Oℓ C2H2g CaOH2aq. TnHep expansion of gas particles in an open container c.
Adding air increases the number of gas particles in the tire. The Gas Pressure Drop App has an intuitive user interface that allows for easy entry of the input data including changes in elevation and selection of valves fittings. Pressure is also exerted by a small sample of gas such as that which is contained in a balloon.
Each result is clearly displayed using expandable. Collisions of gas particles with the walls of the container e. If 10 g of CaC2 are consumed in this reaction how much H2O is needed.
-pressure depends on the frequency of collisions per unit area and on how hard the gas particles strike the container walls-the magnitude of the pressure depends on the chemical identity of the gas-pressure arises when gas particles collide with each other and with the container walls. There are weak forces of attraction between the particles in the gas.

Kolner 9000 5000psi Petrol Engine Pressure Washer 20m Hose Cleaner In 2022 Pressure Washer Washer High Pressure

Have A Future Scientist On Your Hands Well Safety Always Comes First Try Naming These Lab Safety Health And Safety Poster Safety Signs And Symbols

Factors Affecting Gas Pressure Ck 12 Foundation

The 10 Best Gas Pressure Washers Buying Guide Pressure Washer Best Pressure

The 10 Best Gas Pressure Washers Buying Guide Pressure Washer Pressure Best

Rocket Engines Often Feature A Distinctive Pattern Of Diamonds In Their Exhaust These Shock Diamonds Also Known A Fluid Dynamics Rocket Engine Types Of Shock

Factors Affecting Gas Pressure Ck 12 Foundation

The 10 Best Gas Pressure Washers Buying Guide Pressure Washer Washer Pressure Washing

Relating Pressure Volume Amount And Temperature The Ideal Gas Law Chemistry I

What Does Gas Do And Why Does It Always Find Its Way Out This Hands On Resource Shows How Gas Pressure Works Teaching Resources Teaching Teacher Classroom
Pressure And Volume Relationship Of A Gas Boyle S Law Pass My Exams Easy Exam Revision Notes For Gsce Physics

The 10 Best Gas Pressure Washers Buying Guide Best Pressure Washer Pressure Washer Inverter Generators
Pressure And Volume Relationship Of A Gas Boyle S Law Pass My Exams Easy Exam Revision Notes For Gsce Physics


Comments
Post a Comment